The first medication approved by the FDA to treat advanced medullary thyroid cancer (aMTC)
Efficacy
CAPRELSA is a kinase inhibitor indicated for the treatment of symptomatic or progressive medullary thyroid cancer in patients with unresectable locally advanced or metastatic disease. Use CAPRELSA in patients with indolent, asymptomatic or slowly progressing disease only after careful consideration of the treatment-related risks of CAPRELSA.
Clinical Trial Design
ZETA Study
- CAPRELSA was studied in an international, randomized, double-blind, placebo-controlled, phase 3 trial to determine efficacy and safety in adult patients with unresectable locally advanced or metastatic MTC1,2
- One of the largest clinical trials in patients with aMTC
- Patients were followed for progression and survival2
Primary objective: To demonstrate an improvement in progression-free survival (PFS) vs placebo1,2
Secondary objectives: To evaluate objective response rate (ORR), overall survival (OS), and safety1,2
PFS and ORR assessments were based on centralized, independent, blinded review of the imaging data.1
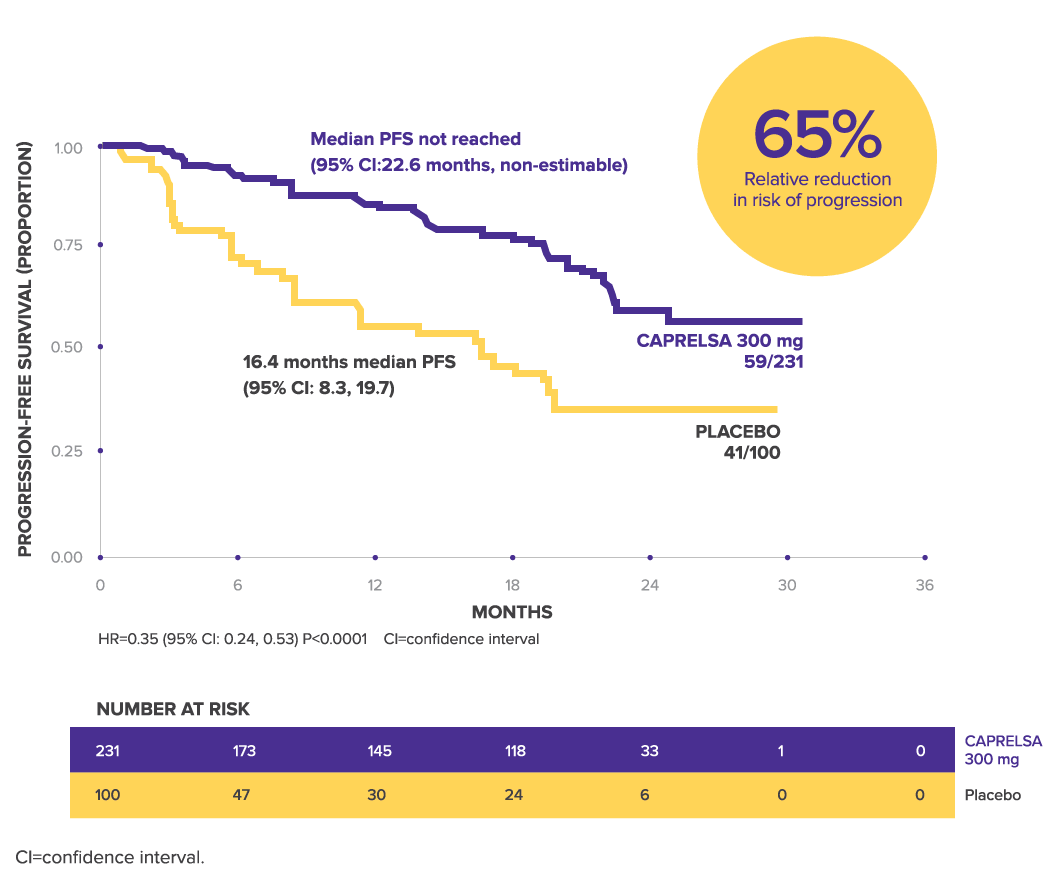
Progression-free Survival
CAPRELSA Significantly Prolonged PFS vs Placebo1,2
Median final OS were similar across both treatment arms.
Comparison of ORR for CAPRELSA and Placebo
44% of CAPRELSA Patients Achieved
Response During Randomized Phase1
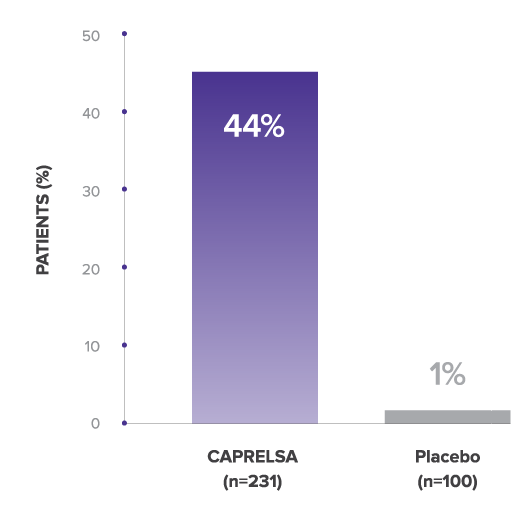
All objective responses were partial responses. No complete responses were observed.1
Mechanism of Action
- In vitro studies have shown that vandetanib inhibits the tyrosine kinase activity of the EGFR and VEGFR families, RET, BRK, TIE2, and members of the EPH receptor and Src kinase families1
- In vitro, vandetanib inhibited EGF-stimulated receptor tyrosine kinase phosphorylation in tumor cells and endothelial cells and VEGF-stimulated tyrosine kinase phosphorylation in endothelial cells1
- In in vivo mouse models of cancer, vandetanib administration reduced tumor cell-induced angiogenesis, tumor vessel permeability, and inhibited tumor growth and metastasis1
EGFR=epidermal growth factor receptor; VEGFR=vascular endothelial growth factor receptor; RET=rearranged during transfection; BRK=breast tumor kinase; TIE2=endothelial-specific receptor tyrosine kinase; EPH=ephrin receptors; Src=non-receptor tyrosine kinases.
References
- CAPRELSA [prescribing information], Genzyme Corporation, Cambridge, MA 02141.
- Wells SA Jr, Robinson BG, Gagel RF, et al. Vandetanib in patients with locally advanced or metastatic medullary thyroid cancer: a randomized, double-blind phase III trial. J Clin Oncol. 2012;30(2):134-141.
Important Safety Information, including Boxed WARNING, for CAPRELSA
WARNING: QT PROLONGATION, TORSADES DE POINTES, AND SUDDEN DEATH
CAPRELSA can prolong the QT interval. Torsades de pointes and sudden death have occurred in patients receiving CAPRELSA. Do not use CAPRELSA in patients with hypocalcemia, hypokalemia, hypomagnesemia, or long QT syndrome. Correct hypocalcemia, hypokalemia and/or hypomagnesemia prior to CAPRELSA administration. Monitor electrolytes periodically. Avoid drugs known to prolong the QT interval. Only prescribers and pharmacies certified with the restricted distribution program are able to prescribe and dispense CAPRELSA.
WARNINGS AND PRECAUTIONS
QT Prolongation and Torsades de Pointes:
- Do not use in patients with congenital long QT syndrome.
- CAPRELSA can prolong the QT interval in a concentration-dependent manner. Torsades de pointes, ventricular tachycardia and sudden deaths have occurred in patients treated with CAPRELSA
- Do not start CAPRELSA treatment in patients whose QTcF interval (corrected QT interval, Fridericia) is greater than 450 ms or who have a history of Torsades de pointes, bradyarrhythmias, or uncompensated heart failure. CAPRELSA has not been studied in patients with ventricular arrhythmias or recent myocardial infarction
- Stop CAPRELSA in patients who develop a QTcF greater than 500 ms until QTcF returns to less than 450 ms. Dosing of CAPRELSA can then be resumed at a reduced dose
- Because of the risk of QT prolongation, obtain an ECG and serum potassium, calcium, magnesium, and thyroid-stimulating hormone (TSH) at baseline, 2-4 weeks and 8-12 weeks after starting treatment with CAPRELSA, and every 3 months thereafter. Following any dose reduction or interruptions greater than 2 weeks, conduct QT assessments as described above
Severe Skin Reactions: Severe skin reactions (including Stevens-Johnson syndrome and Toxic Epidermal Necrolysis), some leading to death, have occurred in patients treated with CAPRELSA. For severe skin reactions, refer patients for urgent medical advice. Systemic therapies e.g., steroids, may be appropriate in such cases and permanent discontinuation of CAPRELSA is recommended. Photosensitivity reactions can occur during CAPRELSA treatment and up to 4 months after treatment discontinuation
Interstitial Lung Disease (ILD): ILD or pneumonitis, including fatalities, has occurred in patients treated with CAPRELSA. Interrupt CAPRELSA for acute or worsening pulmonary symptoms and discontinue CAPRELSA if ILD is confirmed
Ischemic cerebrovascular events: Ischemic cerebrovascular events, including fatalities, occurred in patients treated with CAPRELSA. The safety of resumption of CAPRELSA therapy after resolution of an ischemic cerebrovascular event has not been studied. Discontinue CAPRELSA in patients who experience a severe ischemic cerebrovascular event
Hemorrhage: Serious hemorrhagic events, including fatalities, occurred in patients treated with CAPRELSA. Do not administer CAPRELSA to patients with a recent history of hemoptysis of ≥1/2 teaspoon of red blood. Discontinue CAPRELSA in patients with severe hemorrhage
Heart Failure: Heart failure, including fatalities, occurred in patients treated with CAPRELSA. Monitor for signs and symptoms of heart failure. Consider discontinuation of CAPRELSA in patients with heart failure. Heart failure may not be reversible upon stopping CAPRELSA
Diarrhea: Diarrhea of Grade 3 or greater severity occurred in patients receiving CAPRELSA. If diarrhea occurs, carefully monitor serum electrolytes and ECGs to enable early detection of QT prolongation resulting from dehydration. Interrupt CAPRELSA for severe diarrhea and upon improvement resume CAPRELSA at a reduced dose
Hypothyroidism: Increased dosing of thyroid replacement therapy was required in 49% of CAPRELSA-treated patients. Obtain TSH at baseline, at 2-4 weeks and 8-12 weeks after starting treatment with CAPRELSA, and every 3 months thereafter. If signs or symptoms of hypothyroidism occur, examine thyroid hormone levels and adjust thyroid replacement therapy accordingly
Hypertension: Hypertension, including hypertensive crisis, has occurred in patients treated with CAPRELSA. Monitor all patients for hypertension. Dose reduction or interruption for hypertension may be necessary. If hypertension cannot be controlled, do not resume CAPRELSA
Reversible posterior leukoencephalopathy syndrome (RPLS): RPLS has occurred in patients treated with CAPRELSA. Consider this syndrome in any patient presenting with seizures, headache, visual disturbances, confusion or altered mental function. In clinical studies, three of four patients who developed RPLS while taking CAPRELSA also had hypertension. Discontinue CAPRELSA treatment in patients with RPLS
Drug Interactions: Avoid administration of CAPRELSA with anti-arrhythmic drugs and other drugs that may prolong the QT interval
Renal Failure: Renal failure has occurred in patients treated with CAPRELSA. Vandetanib exposure is increased in patients with impaired renal function. Reduce the starting dose to 200 mg in patients with moderate renal impairment and monitor the QT interval closely. Vandetanib is not recommended for use in patients with severe renal impairment (clearance below 30 mL/min). There is no information available for patients with end-stage renal disease requiring dialysis
Hepatic Impairment: CAPRELSA is not recommended for patients with moderate and severe hepatic impairment, as safety and efficacy have not been established
Impaired Wound Healing: Impaired wound healing has occurred in patients treated with CAPRELSA. Withhold for at least 1 month prior to elective surgery. Do not administer CAPRELSA for at least 2 weeks following major surgery and until adequate wound healing. The safety of resumption of treatment with CAPRELSA after resolution of wound healing complications has not been established.
Embryo-Fetal Toxicity: CAPRELSA can cause fetal harm when administered to a pregnant woman. Advise females of reproductive potential to use effective contraception during treatment with CAPRELSA and for 4 months after the last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with CAPRELSA and for 4 months after the last dose.
CAPRELSA REMS Program: Because of the risks of QT prolongation, Torsades de pointes, and sudden death, CAPRELSA is available only through the CAPRELSA REMS Program. Only prescribers and pharmacies certified with the restricted distribution program are able to prescribe and dispense CAPRELSA. To learn about the specific REMS requirements and to enroll in the CAPRELSA REMS Program, call 1-800-817-2722 or visit www.caprelsarems.com
ADVERSE REACTIONS
The most commonly reported adverse drug reactions (>20%) seen with CAPRELSA and with a between arm difference of ≥5% are diarrhea/colitis (57%), rash (53%), acneiform dermatitis (35%), hypertension (33%), nausea (33%), headache (26%), upper respiratory tract infections (23%), decreased appetite (21%), and abdominal pain (21%).
INDICATIONS AND USAGE
CAPRELSA is a kinase inhibitor indicated for the treatment of symptomatic or progressive medullary thyroid cancer in patients with unresectable locally advanced or metastatic disease.
Use CAPRELSA in patients with indolent, asymptomatic or slowly progressing disease only after careful consideration of the treatment related risks of CAPRELSA.
Please see full Prescribing Information for CAPRELSA, including Boxed WARNING.
